
Alphabetical Index
Chemical Composition of Steels
Keyword Search
Steel Names
Alloyed Steels
Carbon Steels
Cast Irons
Chromium Steels
Cold Work Tool Steels
Creep Resistant Steels
Hot Work Tool Steels
Molybdenum Steels
PM steels
Stainless Steels
Structural Steels
Tool Steels
Vanadium Steels
White Cast Irons
M2C Carbides
M3C Carbides
M7C3 Carbides
M23C6 Carbides
MC Carbides
Light Microscopy
EDS/WDS Microanalysis
Scanning Electron Microscopy
Transmission Electron Microscopy
X-Ray Diffraction
Help
Contact Us
Home
Precipitates in ferritic steels - Demo record
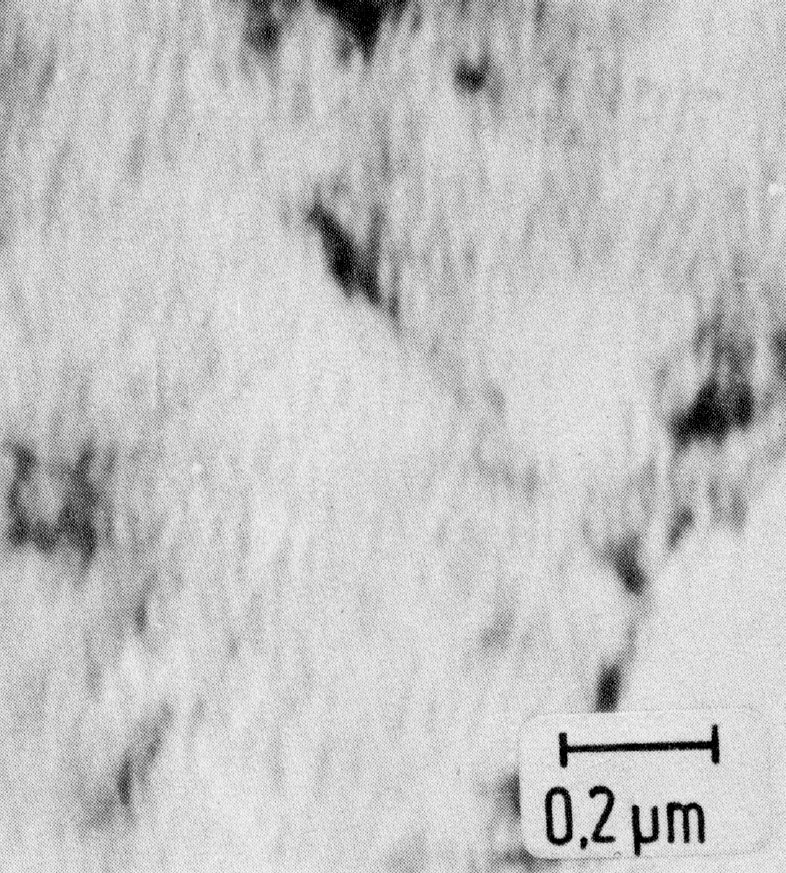
Figure 6: Dissociation of ferrite, thin foil. Scale bar: 0.2 µm.
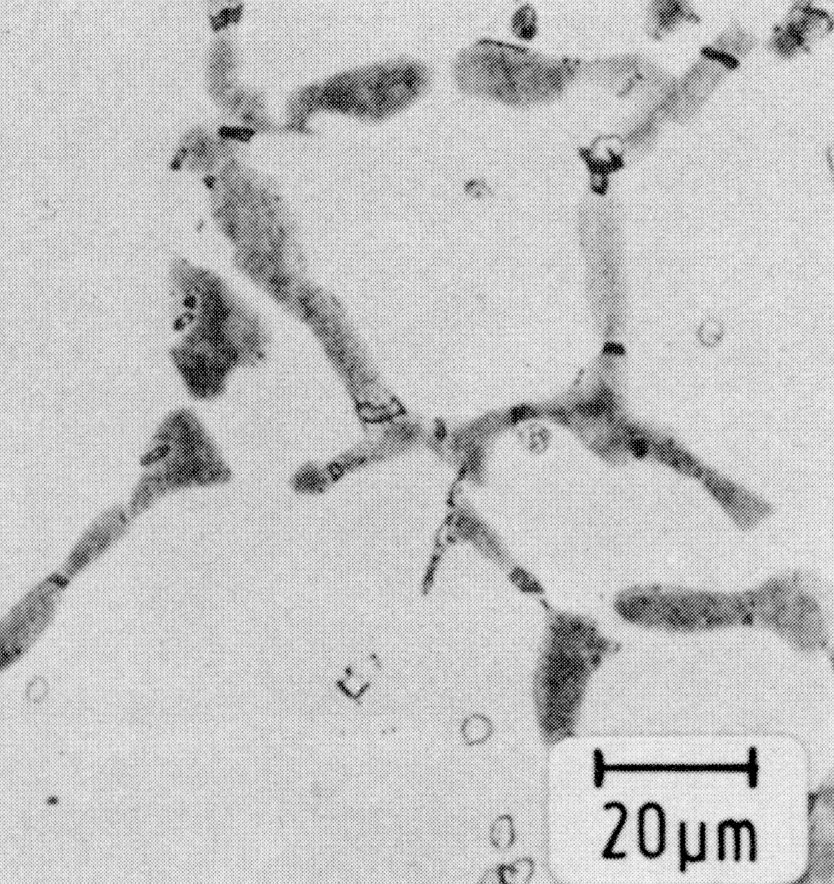
Figure 7: Sigma-phase, light micrograph. Scale bar: 20 µm.

Figure 8: Xsi-phase, light micrograph. Scale bar: 20 µm.
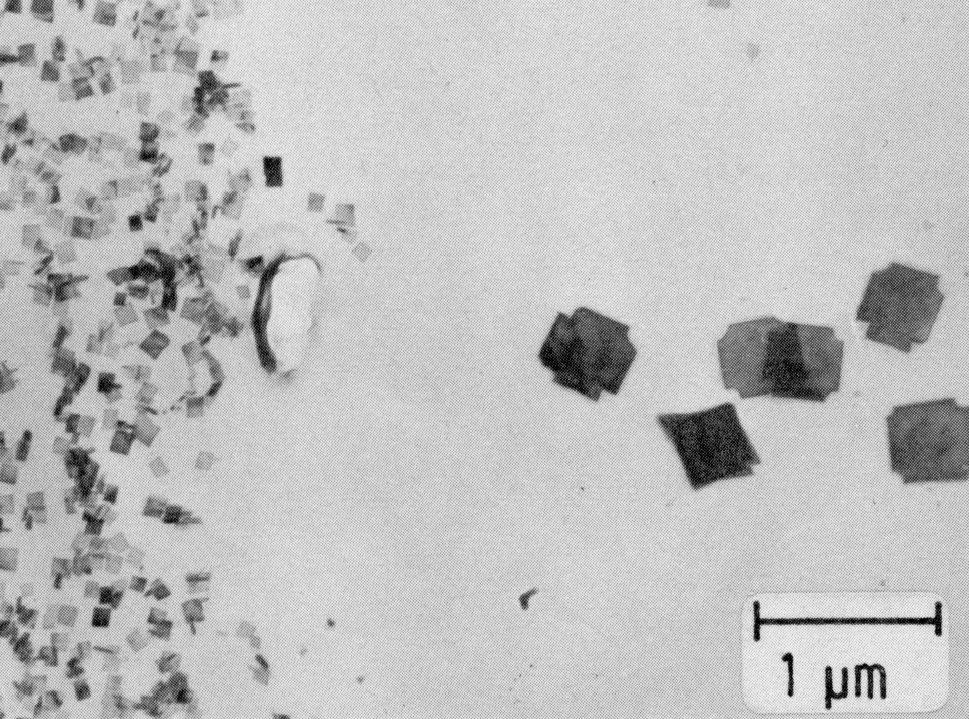
Figure 9: Ti(C,N), extraction replica. Scale bar: 1 µm.
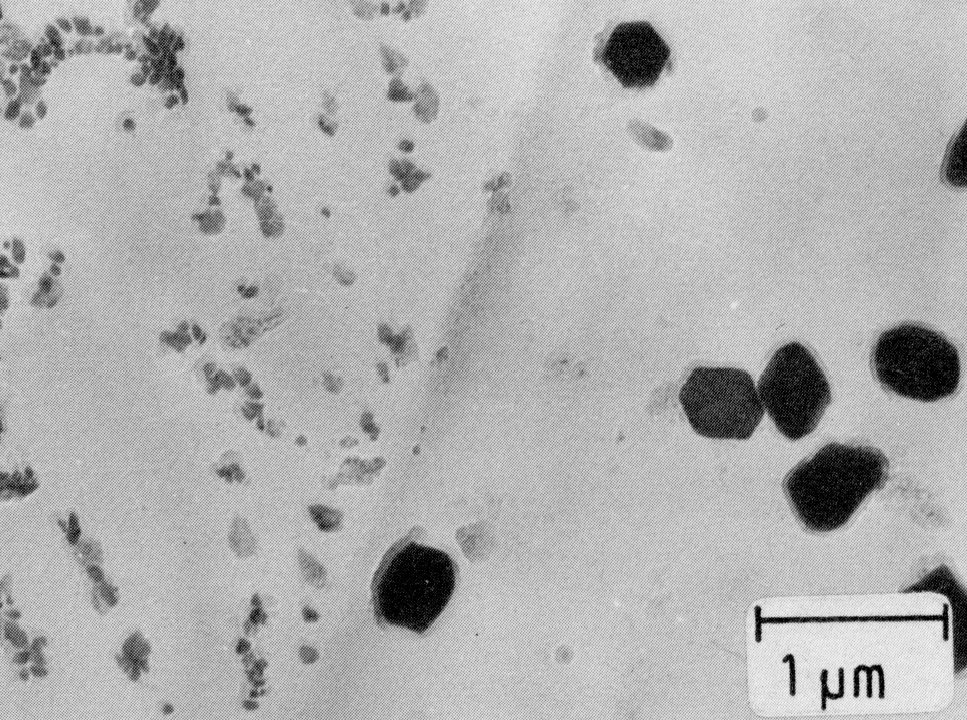
Figure 10: Nb(C,N), extraction replica. Scale bar: 1 µm.

Figure 11: AlN, M23C6 Sigma-phase, light micrograph. Scale bar: 50 µm.

Figure 12: Cr2N, light micrograph. Scale bar: 20 µm.
Carbide name: See the text
Record No.: 1493
Carbide formula: See the text
Carbide type: See the text
Carbide composition in weight %: No data
Image type: TEM, LM
Steel name: Ferritic steel
Mat.No. (Wr.Nr.) designation: No data
DIN designation: No data
AISI/SAE/ASTM designation: No data
Other designation: No data
Steel group: Ferritic steels
Steel composition in weight %: No data
Heat treatment/condition: See the text
Note: The ferritic steels cool from the melting point to room temperature without any transformation. The condition of the homogeneous ferritic microstructure formed in this way is a constriction of the gamma-region in the phase diagram. The increasing restriction and suppression of the range of existence of the gamma-phase, or austenite, can be achieved by the addition of various alloying elements which stabilize the ferritic state; these are, amongst others, chromium, silicon, molybdenum, niobium, vanadium, aluminium and titanium. With the loss of the gamma-alpha transformation the possibility of hardening by martensite formation is also forfeited.
The commonest alloying addition used industrially for attaining a purely ferritic microstructure is chromium. The chromium content of the ferritic steels ranges from about 6 to 28%. The alloys with a high chromium content and considerably reduced carbon contents are known as super-ferrites. Increasing carbon contents inhibit the ferrite-forming effect and lead under certain circumstances to the so-called semi-ferritic steels, which have properties between those of the tempering steels and the pure ferritic steels with regard to hardness and toughness.
Compared with the hardenable steel types, the ferritic steels have, on account of their chromium content, a greater corrosion resistance, which may be further increased by molybdenum contents up to 2%. With their good resistance to acids, such steels are often used in chemical plants. Additions of about 0.5 to 1.5% Si and/or Al improve the scaling resistance and reduce the sensitivity to gases containing sulphur, so that such alloys tend to be used as heat-resistant materials.
In iron-chromium steels high corrosion resistance and good technological properties are to be expected only if the sum of the carbon and nitrogen contents is very low. Since the smelting of such alloys is difficult, some processes require the binding of carbon and nitrogen by the powerful carbide and nitride-forming elements titanium or niobium. The carbonitrides then formed counteract the marked tendency to grain growth and the associated loss of toughness. They exert a stabilizing influence on intergranular corrosion, but may, in the form of finely dispersed particles and grain boundary precipitates, have a detrimental effect on the mechanical properties. Therefore nickel is often added for compensation in an amount which, as far as possible, does not yet lead to the formation of a mixed microstructure of ferrite and austenite, but favourably influences the corrosion properties in addition.
In nickel-free iron-chromium alloys, two precipitation processes occur in different temperature ranges below the saturation limit (950 °C):
Below 550 °C the 475 °C embrittlement is observed, consisting of a single-phase decomposition with the formation of submicroscopically fine precipitates (10 nm) of a chromium-rich phase which may contain up to 80% Cr (Fig. 6).
Above 550 °C the Sigma-phase Fe(Cr,Mo) (Fig. 7), which also causes embrittlement, and/or the molybdenum-rich Xsi-phase Fe36Cr12Mo10 (Fig. 8) are formed, depending upon the conditions of the heat treatment.
The range of stability of these two phases is extended by nickel and also by titanium, which may be used for stabilizing. Niobium, also used for binding carbon and nitrogen, does not have this effect. After a solution treatment at about 1050 °C, the carbonitrides of titanium and niobium are still in the form of more or less fine precipitates (Fig. 9 and Fig. 10).
Higher nickel contents lead to the formation of austenite at temperatures above 850 °C. This favours the formation of the Sigma-phase due to chromium enrichment in the ferrite.
The precipitation of Sigma and Xsi-phase begins on the grain boundaries and continues with increasing aging time in the ferrite grain in the form of polyhedra (Figs. 2 and 3). Small amounts of titanium and niobium may be taken up by the iron-chromium-molybdenum phases.
The formation of the above-mentioned intermetallic compounds is determined to a large extent by the chromium/molybdenum ratio. In steels with 20% Cr the Xsi-phase predominates, in those with 28% Cr the Sigma-phase, on account of the higher chromium/molybdenum ratio.
Higher aluminium contents may lead to the formation of coarse aluminium nitrides after long times in the temperature range from 600 to 900 °C. Such precipitates, together with other phases occurring in these steels, such as M23C6 and the Sigma-phase, are shown in Fig. 11. Nitriding at high temperatures may lead to the precipitation of hexagonal Cr2N, which may be recognized by its characteristic needleshaped form (Fig. 12).
Links: No data
Reference: Not shown in this demo version.